|
The recipient of a Bachelor of Science in Biomedical Science from the City College of New York, Wayne Macfadden MD is a former assistant professor of psychiatry at the University of Pennsylvania School of Medicine. Dr. Wayne Macfadden has provided pharmaceutical therapy for patients with various mental illnesses and substance use disorders.
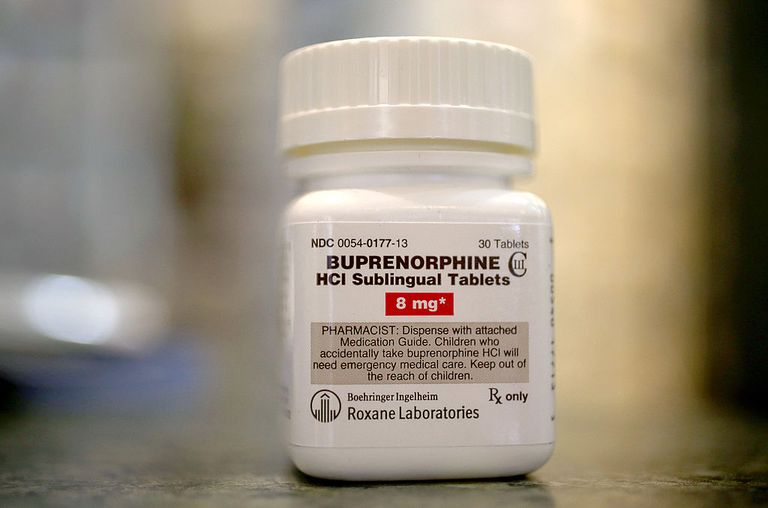
A compound that acts as an opioid agonist, buprenorphine works differently from full opioid agonists by activating opioid receptors less strongly, thereby making it a partial opioid agonist. Buprenorphine can bring relief to a patient undergoing opioid withdrawal, making it useful in the treatment of opiate use disorder. In the United States, buprenorphine is usually administered in the form of tablets or films. The film is administered orally under the tongue or between the gums and cheek, and is often sold under the brand name Suboxone. In order to prevent the injection of the medication, which may have harmful consequences, the film contains buprenorphine combined with naloxone, a narcotic opioid antagonist that will rapidly reduce the effect of the product if injected. When the film is administered orally, withdrawal should not occur. In addition, buprenorphine is also available in implantable formulations as well as an extended-release formulation, which may result in an increase in compliance to treatment, possibly with better outcomes. Because it has less effect on respiratory depression, buprenorphine has a lower risk of drug overdose compared to other opiate medications. Its therapeutic dose can usually be determined within a few days.
0 Comments
The recipient of an MD degree from the State University Of New York – Buffalo School of Medicine, Ft. Wayne Macfadden is a psychiatrist and former director at Substance Abuse Treatment Services. Dr. Wayne Macfadden has treated various psychiatric disorders and has also used buprenorphine in the medication-assisted treatment of opiate use disorder.
Approved in 2002 by the United States Food and Drug Administration, buprenorphine is the first medication marked eligible for prescription by certified physicians using the Drug Addiction Treatment Act. Since 2010, a sublingual film of buprenorphine has been made available for use in pharmacotherapy. In addition, the Comprehensive Addiction and Recovery Act temporarily expands eligibility of buprenorphine-based drug prescription for medication-assisted treatments to qualifying physician assistants and nurse practitioners through October 1, 2021. A recent approval of buprenorphine implant therapy was made by the FDA in May 2016, when a six-month-duration subdermal buprenorphine implant therapy was approved. In November 2017, a once-monthly buprenorphine injection was also approved. The purpose of these longer-term administrations is to eliminate the treatment barrier of daily dosing for patients stabilized on buprenorphine. |
AuthorWayne Macfadden MD: Expert in both Clinical and Research Psychiatry. Archives
March 2024
Categories |

 RSS Feed
RSS Feed
